Eversense is a smart pill-sized implantable Continuous Glucose Monitoring (CGM) system by Senseonics, a medical device company in Germantown, Maryland. The Eversense CGM system is based on fluorescence sensing technology.

Three core products of the company are 90-day implantable sensor, a removable and rechargeable smart transmitter, and a mobile app that eliminates the need for separate receiver.
The sensor which is encased in a bio-compatible material works by measuring interstitial fluid glucose levels in adults with diabetes. The company promises the sensor to be highly accurate and stable up to 90 days.
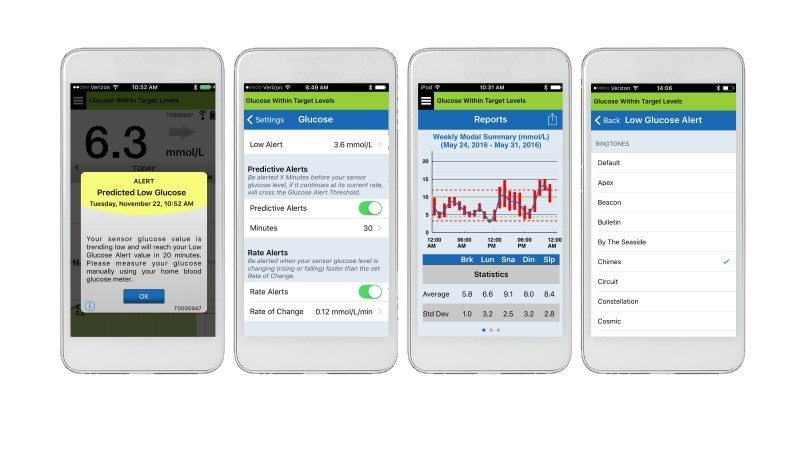
The measurement and display of glucose values takes place without human intervention. The smart transmitter is worn over a patient’s sensor and wirelessly powers it to activate the transfer of glucose measurements. After receiving glucose data from the sensor, it calculates the glucose level and transmits it via Bluetooth to the Eversense mobile app. It receives readings every 5 minutes.

The company’s proprietary Eversense Data Management System is a browser-based application that enables patients to upload glucose readings from Eversense Mobile App. It lets patients send predictive alerts to their smartphone before they reach abnormally low or high glucose level. The CGM system provides a “moving” picture of how a patient’s glucose levels change over time without continuous samples from fingersticks. However, Senseonics recommends using its system as an adjunctive device to complement, not replace, information obtained from standard home blood glucose monitoring devices.